What Is Healthier Grass Feed Ir Organic Beef
- Review
- Open Access
- Published:
A review of fatty acid profiles and antioxidant content in grass-fed and grain-fed beef
Nutrition Periodical book nine, Commodity number:10 (2010) Cite this article
Abstruse
Growing consumer interest in grass-fed beef products has raised a number of questions with regard to the perceived differences in nutritional quality between grass-fed and grain-fed cattle. Research spanning three decades suggests that grass-based diets can significantly improve the fat acid (FA) composition and antioxidant content of beef, albeit with variable impacts on overall palatability. Grass-based diets have been shown to enhance total conjugated linoleic acrid (CLA) (C18:2) isomers, trans vaccenic acid (TVA) (C18:1 t11), a precursor to CLA, and omega-3 (n-3) FAs on a chiliad/thousand fat footing. While the overall concentration of total SFAs is not different between feeding regimens, grass-finished beef tends toward a higher proportion of cholesterol neutral stearic FA (C18:0), and less cholesterol-elevating SFAs such as myristic (C14:0) and palmitic (C16:0) FAs. Several studies suggest that grass-based diets elevate precursors for Vitamin A and East, also as cancer fighting antioxidants such as glutathione (GT) and superoxide dismutase (SOD) activeness as compared to grain-fed contemporaries. Fat witting consumers will likewise prefer the overall lower fat content of a grass-fed beef production. Nevertheless, consumers should be aware that the differences in FA content will as well requite grass-fed beefiness a distinct grass flavor and unique cooking qualities that should be considered when making the transition from grain-fed beef. In improver, the fat from grass-finished beef may take a yellow advent from the elevated carotenoid content (precursor to Vitamin A). Information technology is as well noted that grain-fed beef consumers may achieve similar intakes of both n-iii and CLA through the consumption of higher fat grain-fed portions.
Review Contents
- one.
Introduction
- two.
Fatty acid profile in grass-fed beef
- iii.
Impact of grass-finishing on omega-3 fatty acids
- iv.
Impact of grass-finishing on conjugated linoleic acid (CLA) and trans-vaccenic acid (TVA)
- five.
Impact of grass-finishing on β-carotenes/carotenoids
- half dozen.
Affect of grass-finishing on α-tocopherol
- 7.
Affect of grass-finishing on GT & SOD activity
- 8.
Bear on of grass-finishing on flavor and palatability
- nine.
Conclusion
- 10.
References
Introduction
There is considerable back up amongst the nutritional communities for the diet-heart (lipid) hypothesis, the idea that an imbalance of dietary cholesterol and fats are the primary cause of atherosclerosis and cardiovascular disease (CVD) [1]. Health professionals globe-wide recommend a reduction in the overall consumption of SFAs, trans-fatty acids (TAs) and cholesterol, while emphasizing the need to increase intake of northward-3 polyunsaturated fats [one, ii]. Such broad sweeping nutritional recommendations with regard to fat consumption are largely due to epidemiologic studies showing strong positive correlations betwixt intake of SFA and the incidence of CVD, a condition believed to result from the concomitant ascension in serum low-density-lipoprotein (LDL) cholesterol as SFA intake increases [iii, 4]. For example, it is generally accustomed that for every one% increment in energy from SFA, LDL cholesterol levels reportedly increase by 1.3 to 1.7 mg/dL (0.034 to 0.044 mmol/L) [five–vii].
Broad promotion of this correlative information spurred an anti-SFA entrada that reduced consumption of dietary fats, including most animal proteins such as meat, dairy products and eggs over the terminal three decades [8], indicted on their relatively high SFA and cholesterol content. However, more recent lipid inquiry would advise that not all SFAs have the aforementioned impact on serum cholesterol. For instance, lauric acid (C12:0) and myristic acid (C14:0), have a greater total cholesterol raising issue than palmitic acid (C16:0), whereas stearic acid (C18:0) has a neutral outcome on the concentration of full serum cholesterol, including no apparent impact on either LDL or HDL. Lauric acid increases total serum cholesterol, although it too decreases the ratio of full cholesterol:HDL considering of a preferential increase in HDL cholesterol [v, 7, 9]. Thus, the individual fatty acrid profiles tend to be more instructive than broad lipid classifications with respect to subsequent impacts on serum cholesterol, and should therefore be considered when making dietary recommendations for the prevention of CVD.
Conspicuously the lipid hypothesis has had broad sweeping impacts; not only on the way we eat, but also on the mode food is produced on-farm. Indeed, changes in animal breeding and genetics have resulted in an overall leaner beef production[10]. Preliminary examination of diets containing today's bacteria beefiness has shown a reduction in serum cholesterol, provided that beefiness consumption is limited to a iii ounce portion devoid of all external fat [eleven]. O'Dea's work was the commencement of several studies to bear witness today's bacteria beef products tin reduce plasma LDL concentrations in both normal and hyper-cholesterolemic subjects, theoretically reducing risk of CVD [12–xv].
Beyond changes in genetics, some producers have also contradistinct their feeding practices whereby reducing or eliminating grain from the ruminant nutrition, producing a production referred to equally "grass-fed" or "grass-finished". Historically, most of the beefiness produced until the 1940's was from cattle finished on grass. During the 1950's, considerable research was done to improve the efficiency of beef product, giving birth to the feedlot industry where high free energy grains are fed to cattle as ways to decrease days on feed and improve marbling (intramuscular fat: Imf). In addition, U.Due south. consumers accept grown accustomed to the taste of grain-fed beef, generally preferring the flavor and overall palatability afforded by the college energy grain ration[16]. Even so, changes in consumer demand, coupled with new research on the effect of feed on food content, take a number of producers returning to the pastoral approach to beef production despite the inherent inefficiencies.
Enquiry spanning three decades suggests that grass-only diets tin significantly alter the fatty acid limerick and ameliorate the overall antioxidant content of beef. It is the intent of this review, to synthesize and summarize the information currently available to substantiate an enhanced nutrient claim for grass-fed beef products also as to hash out the effects these specific nutrients accept on human being health.
Review of fatty acid profiles in grass-fed beef
Cherry meat, regardless of feeding regimen, is nutrient dense and regarded equally an important source of essential amino acids, vitamins A, Bhalf-dozen, B12, D, E, and minerals, including iron, zinc and selenium [17, eighteen]. Forth with these of import nutrients, meat consumers also ingest a number of fats which are an of import source of energy and facilitate the absorption of fat-soluble vitamins including A, D, E and K. Co-ordinate to the ADA, brute fats contribute approximately 60% of the SFA in the American diet, most of which are palmitic acid (C16:0) and stearic acrid (C18:0). Stearic acid has been shown to take no net impact on serum cholesterol concentrations in humans[17, 19]. In addition, 30% of the FA content in conventionally produced beefiness is composed of oleic acrid (C18:one) [twenty], a monounsaturated FA (MUFA) that elicits a cholesterol-lowering effect among other healthful attributes including a reduced run a risk of stroke and a pregnant subtract in both systolic and diastolic blood force per unit area in susceptible populations [21].
Be that as it may, changes in finishing diets of conventional cattle can alter the lipid profile in such a mode as to improve upon this nutritional package. Although there are genetic, age related and gender differences among the various meat producing species with respect to lipid profiles and ratios, the outcome of brute diet is quite pregnant [22]. Regardless of the genetic makeup, gender, age, species or geographic location, straight contrasts between grass and grain rations consistently demonstrate pregnant differences in the overall fatty acrid profile and antioxidant content establish in the lipid depots and body tissues [22–24].
Table 1 summarizes the saturated fatty acid analysis for a number of studies whose objectives were to contrast the lipid profiles of cattle fed either a grain or grass diets [25–31]. This table is express to those studies utilizing the longissimus dorsi (loin eye), thereby standardizing the contrasts to like cuts within the carcass and limits the comparisons to cattle betwixt xx and 30 months of age. Unfortunately, not all studies study information in like units of measure (i.e., g/yard of fatty acrid), so direct comparisons between studies are not possible.
Table 1 reports that grass finished cattle are typically lower in total fat equally compared to grain-fed contemporaries. Interestingly, in that location is no consequent difference in full SFA content between these ii feeding regimens. Those SFA's considered to be more than detrimental to serum cholesterol levels, i.e., myristic (C14:0) and palmitic (C16:0), were higher in grain-fed beef as compared to grass-fed contemporaries in sixty% of the studies reviewed. Grass finished meat contains elevated concentrations of stearic acid (C18:0), the only saturated fatty acid with a net neutral impact on serum cholesterol. Thus, grass finished beef tends to produce a more than favorable SFA limerick although fiddling is known of how grass-finished beefiness would ultimately impact serum cholesterol levels in hyper-cholesterolemic patients as compared to a grain-fed beefiness.
Like SFA intake, dietary cholesterol consumption has also become an important issue to consumers. Interestingly, beef's cholesterol content is similar to other meats (beefiness 73; pork 79; lamb 85; craven 76; and turkey 83 mg/100 thousand) [32], and can therefore be used interchangeably with white meats to reduce serum cholesterol levels in hyper-cholesterolemic individuals[11, 33]. Studies take shown that breed, nutrition and sex do non affect the cholesterol concentration of bovine skeletal muscle, rather cholesterol content is highly correlated to IMF concentrations[34]. As IMF levels rise, so goes cholesterol concentrations per gram of tissue [35]. Because pasture raised beef is lower in overall fatty [24–27, 30], particularly with respect to marbling or Imf [26, 36], it would seem to follow that grass-finished beef would be lower in overall cholesterol content although the data is very limited. Garcia et al (2008) report 40.3 and 45.eight grams of cholesterol/100 grams of tissue in pastured and grain-fed steers, respectively (P < 0.001) [24].
Interestingly, grain-fed beef consistently produces higher concentrations of MUFAs as compared to grass-fed beef, which include FAs such as oleic acid (C18:ane cis-9), the primary MUFA in beef. A number of epidemiological studies comparing disease rates in different countries have suggested an inverse association between MUFA intake and mortality rates to CVD [3, 21]. Nonetheless, grass-fed beef provides a higher concentration of TVA (C18:ane t11), an important MUFA for de novo synthesis of conjugated linoleic acid (CLA: C18:2 c-9, t-11), a potent anti-carcinogen that is synthesized within the torso tissues [37]. Specific information relative to the wellness benefits of CLA and its biochemistry will be detailed later.
The important polyunsaturated fatty acids (PUFAs) in conventional beef are linoleic acid (C18:two), alpha-linolenic acrid (C18:iii), described as the essential FAs, and the long-chain fatty acids including arachidonic acid (C20:4), eicosapentaenoic acid (C20:five), docosanpetaenoic acid (C22:v) and docosahexaenoic acid (C22:6) [38]. The significance of diet on fatty acid composition is clearly demonstrated when profiles are examined by omega vi (due north-6) and omega three (n-3) families. Tabular array 2 shows no significant change to the overall concentration of n-six FAs betwixt feeding regimens, although grass-fed beef consistently shows a college concentrations of n-iii FAs as compared to grain-fed contemporaries, creating a more than favorable n-half-dozen:north-3 ratio. There are a number of studies that study positive furnishings of improved n-3 intake on CVD and other health related problems discussed in more detail in the adjacent department.
Review of Omega-iii: Omega-6 fatty acid content in grass-fed beef
At that place are two essential fatty acids (EFAs) in human nutrition: α-linolenic acid (αLA), an omega-3 fatty acrid; and linoleic acid (LA), an omega-6 fat acid. The man body cannot synthesize essential fat acids, even so they are critical to human being health; for this reason, EFAs must be obtained from food. Both αLA and LA are polyunsaturated and serve as precursors of other important compounds. For example, αLA is the precursor for the omega-3 pathway. Also, LA is the parent fatty acid in the omega-6 pathway. Omega-3 (n-3) and omega-6 (north-6) fatty acids are two separate distinct families, yet they are synthesized by some of the aforementioned enzymes; specifically, delta-5-desaturase and delta-6-desaturase. Excess of one family unit of FAs can interfere with the metabolism of the other, reducing its incorporation into tissue lipids and altering their overall biological effects [39]. Figure 1 depicts a schematic of n-6 and n-3 metabolism and elongation within the body [40].
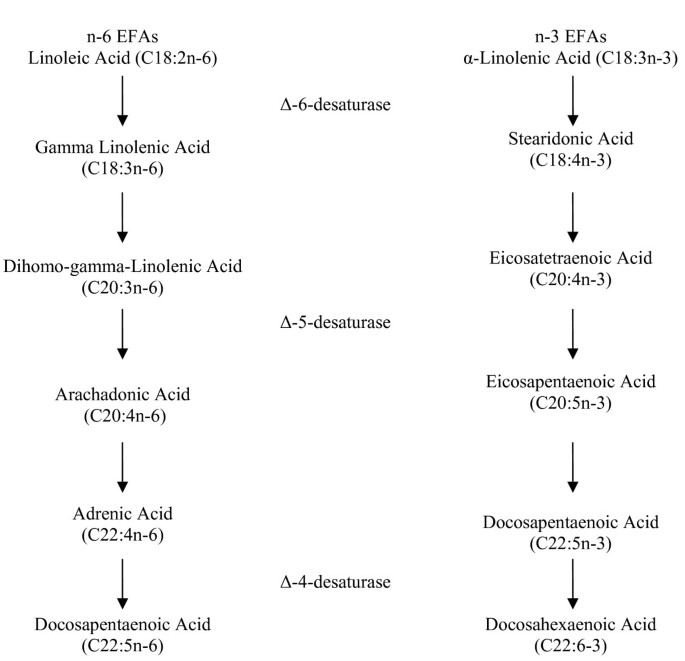
Linoleic (C18:2n-6) and α-Linolenic (C18:3n-iii) Acid metabolism and elongation. (Adjusted from Simopoulos et al., 1991)
A good for you nutrition should consist of roughly 1 to four times more omega-6 fatty acids than omega-three fat acids. The typical American diet tends to comprise xi to 30 times more omega -6 fatty acids than omega -3, a miracle that has been hypothesized every bit a meaning cistron in the rising rate of inflammatory disorders in the U.s.a.[twoscore]. Table 2 shows pregnant differences in north-6:n-three ratios between grass-fed and grain-fed beef, with and overall average of 1.53 and 7.65 for grass-fed and grain-fed, respectively, for all studies reported in this review.
The major types of omega-iii fatty acids used by the body include: α-linolenic acid (C18:3n-3, αLA), eicosapentaenoic acid (C20:5n-iii, EPA), docosapentaenoic acid (C22:5n-3, DPA), and docosahexaenoic acid (C22:6n-3, DHA). In one case eaten, the body converts αLA to EPA, DPA and DHA, albeit at low efficiency. Studies generally agree that whole torso conversion of αLA to DHA is below 5% in humans, the majority of these long-concatenation FAs are consumed in the diet [41].
The omega-three fat acids were starting time discovered in the early 1970's when Danish physicians observed that Greenland Eskimos had an exceptionally depression incidence of middle illness and arthritis despite the fact that they consumed a diet high in fatty. These early on studies established fish as a rich source of northward-3 fat acids. More recent research has established that EPA and DHA play a crucial office in the prevention of atherosclerosis, heart set on, depression and cancer [twoscore, 42]. In improver, omega-iii consumption reduced the inflammation caused by rheumatoid arthritis [43, 44].
The human being encephalon has a high requirement for DHA; low DHA levels have been linked to low brain serotonin levels, which are connected to an increased tendency for depression and suicide. Several studies have established a correlation between depression levels of omega -three fatty acids and depression. Loftier consumption of omega-3 FAs is typically associated with a lower incidence of depression, a decreased prevalence of historic period-related retentiveness loss and a lower take a chance of developing Alzheimer'due south disease [45–51].
The National Institutes of Health has published recommended daily intakes of FAs; specific recommendations include 650 mg of EPA and DHA, 2.22 one thousand/day of αLA and 4.44 g/day of LA. Even so, the Plant of Medicine has recommended DRI (dietary reference intake) for LA (omega-6) at 12 to 17 g and αLA (omega-three) at 1.one to 1.6 g for developed women and men, respectively. Although seafood is the major dietary source of n-iii fatty acids, a recent fatty acid intake survey indicated that cherry-red meat also serves as a significant source of n-3 fatty acids for some populations [52].
Sinclair and co-workers were the first to show that beef consumption increased serum concentrations of a number of n-three fat acids including, EPA, DPA and DHA in humans [40]. Likewise, in that location are a number of studies that have been conducted with livestock which report similar findings, i.eastward., animals that consume rations high in forerunner lipids produce a meat product higher in the essential fat acids [53, 54]. For instance, cattle fed primarily grass significantly increased the omega-3 content of the meat and as well produced a more favorable omega-vi to omega-3 ratio than grain-fed beef [46, 55–57].
Table 2 shows the effect of ration on polyunsaturated fatty acid composition from a number of recent studies that dissimilarity grass-based rations to conventional grain feeding regimens [24–28, xxx, 31]. Grass-based diets resulted in significantly higher levels of omega-3 within the lipid fraction of the meat, while omega-vi levels were left unchanged. In fact, as the concentration of grain is increased in the grass-based diet, the concentration of n-3 FAs decreases in a linear fashion. Grass-finished beef consistently produces a higher concentration of n-3 FAs (without effecting n-6 FA content), resulting in a more favorable n-vi:n-three ratio.
The amount of total lipid (fatty) constitute in a serving of meat is highly dependent upon the feeding regimen as demonstrated in Tables 1 and 2. Fatty volition likewise vary by cutting, as non all locations of the carcass will deposit fat to the same degree. Genetics also play a role in lipid metabolism creating pregnant breed effects. Still, the upshot of feeding regimen is a very powerful determinant of fat acid composition.
Review of conjugated linoleic acid (CLA) and transvaccenic acrid (TVA) in grass-fed beef
Conjugated linoleic acids make up a group of polyunsaturated FAs institute in meat and milk from ruminant animals and be as a general mixture of conjugated isomers of LA. Of the many isomers identified, the cis-nine, trans-11 CLA isomer (as well referred to every bit rumenic acrid or RA) accounts for upward to lxxx-xc% of the full CLA in ruminant products [58]. Naturally occurring CLAs originate from two sources: bacterial isomerization and/or biohydrogenation of polyunsaturated fatty acids (PUFA) in the rumen and the desaturation of trans-fatty acids in the adipose tissue and mammary gland [59, 60].
Microbial biohydrogenation of LA and αLA by an anaerobic rumen bacterium Butyrivibrio fibrisolvens is highly dependent on rumen pH [61]. Grain consumption decreases rumen pH, reducing B. fibrisolven activity, conversely grass-based diets provide for a more than favorable rumen environment for subsequent bacterial synthesis [62]. Rumen pH may help to explicate the credible differences in CLA content betwixt grain and grass-finished meat products (run across Table two). De novo synthesis of CLA from xit-C18:i TVA has been documented in rodents, dairy cows and humans. Studies advise a linear increase in CLA synthesis as the TVA content of the diet increased in human subjects [63]. The rate of conversion of TVA to CLA has been estimated to range from 5 to 12% in rodents to nineteen to thirty% in humans[64]. True dietary intake of CLA should therefore consider native 9c11t-C18:2 (actual CLA) as well as the 11t-C18:1 (potential CLA) content of foods [65, 66]. Effigy 2 portrays de novo synthesis pathways of CLA from TVA [37].
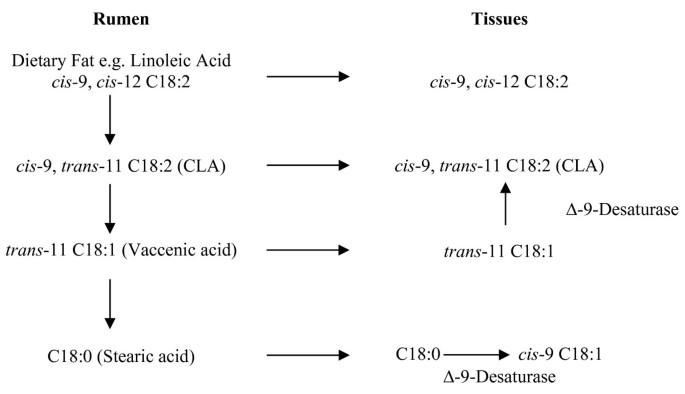
De novo synthesis of CLA from 11t-C18:1 vaccenic acid. (Adapted from Bauman et al., 1999)
Natural augmentation of CLA cixtxi and TVA within the lipid fraction of beef products can exist accomplished through diets rich in grass and lush greenish forages. While precursors tin can be constitute in both grains and lush green forages, grass-fed ruminant species have been shown to produce 2 to three times more CLA than ruminants fed in solitude on high grain diets, largely due to a more favorable rumen pH [34, 56, 57, 67] (come across Tabular array 2).
The impact of feeding practices becomes even more axiomatic in light of recent reports from Canada which suggests a shift in the predominate trans C18:one isomer in grain-fed beef. Dugan et al (2007) reported that the major trans isomer in beef produced from a 73% barley grain diet is 10t-18:1 (2.13% of total lipid) rather than 11t-18:one (TVA) (0.77% of total lipid), a finding that is not especially favorable considering the data that would support a negative touch on of xt-18:ane on LDL cholesterol and CVD [68, 69].
Over the past two decades numerous studies have shown significant wellness benefits owing to the deportment of CLA, equally demonstrated past experimental animal models, including actions to reduce carcinogenesis, atherosclerosis, and onset of diabetes [70–72]. Conjugated linoleic acid has too been reported to attune trunk composition by reducing the accumulation of adipose tissue in a diversity of species including mice, rats, pigs, and now humans [73–76]. These changes in body composition occur at ultra high doses of CLA, dosages that can only exist attained through constructed supplementation that may also produce sick side-effects, such every bit gastrointestinal upset, adverse changes to glucose/insulin metabolism and compromised liver office [77–81]. A number of fantabulous reviews on CLA and human wellness can be plant in the literature [61, 82–84].
Optimal dietary intake remains to be established for CLA. It has been hypothesized that 95 mg CLA/day is plenty to show positive effects in the reduction of breast cancer in women utilizing epidemiological data linking increased milk consumption with reduced chest cancer[85]. Ha et al. (1989) published a much more conservative approximate stating that 3 g/day CLA is required to promote man wellness benefits[86]. Ritzenthaler et al. (2001) estimated CLA intakes of 620 mg/twenty-four hour period for men and 441 mg/day for women are necessary for cancer prevention[87]. Obviously, all these values represent crude estimates and are mainly based on extrapolated animal data. What is clear is that we as a population do not consume enough CLA in our diets to have a significant affect on cancer prevention or suppression. Reports betoken that Americans consume between 150 to 200 mg/day, Germans consumer slightly more betwixt 300 to 400 mg/day[87], and the Australians seem to be closer to the optimum concentration at 500 to 1000 mg/solar day according to Parodi (1994) [88].
Review of pro-Vitamin A/β-carotene in grass-fed meat
Carotenoids are a family of compounds that are synthesized past higher plants as natural institute pigments. Xanthophylls, carotene and lycopene are responsible for xanthous, orange and red coloring, respectively. Ruminants on high provender rations laissez passer a portion of the ingested carotenoids into the milk and trunk fatty in a fashion that has still to be fully elucidated. Cattle produced under extensive grass-based product systems generally have carcass fatty which is more than yellow than their concentrate-fed counterparts caused past carotenoids from the lush light-green forages. Although yellow carcass fat is negatively regarded in many countries around the world, it is likewise associated with a healthier fat acid profile and a higher antioxidant content [89].
Plant species, harvest methods, and season, all have pregnant impacts on the carotenoid content of forage. In the procedure of making silage, haylage or hay, every bit much equally eighty% of the carotenoid content is destroyed [90]. Further, pregnant seasonal shifts occur in carotenoid content owing to the seasonal nature of establish growth.
Carotenes (mainly β-carotene) are precursors of retinol (Vitamin A), a critical fat-soluble vitamin that is important for normal vision, os growth, reproduction, cell partition, and jail cell differentiation [91]. Specifically, it is responsible for maintaining the surface lining of the eyes and too the lining of the respiratory, urinary, and abdominal tracts. The overall integrity of skin and mucous membranes is maintained by vitamin A, creating a bulwark to bacterial and viral infection [15, 92]. In addition, vitamin A is involved in the regulation of immune role by supporting the production and office of white blood cells [12, xiii].
The current recommended intake of vitamin A is 3,000 to 5,000 IU for men and 2,300 to iv,000 IU for women [93], respectively, which is equivalent to 900 to 1500 μg (micrograms) (Annotation: DRI equally reported past the Constitute of Medicine for not-significant/non-lactating adult females is 700 μg/day and males is 900 μg/twenty-four hours or 2,300 - 3,000 I U (bold conversion of three.33 IU/μg). While there is no RDA (Required Daily Assart) for β-carotene or other pro-vitamin A carotenoids, the Institute of Medicine suggests consuming three mg of β-carotene daily to maintain plasma β-carotene in the range associated with normal part and a lowered chance of chronic diseases (NIH: Office of Dietary Supplements).
The furnishings of grass feeding on beta-carotene content of beef was described past Descalzo et al. (2005) who constitute pasture-fed steers incorporated significantly college amounts of beta-carotene into musculus tissues as compared to grain-fed animals [94]. Concentrations were 0.45 μg/yard and 0.06 μg/m for beef from pasture and grain-fed cattle respectively, demonstrating a 7 fold increase in β-carotene levels for grass-fed beef over the grain-fed contemporaries. Similar information has been reported previously, presumably due to the loftier β-carotene content of fresh grasses every bit compared to cereal grains[38, 55, 95–97]. (see Table 3)
Review of Vitamin Due east/α-tocopherol in grass-fed beef
Vitamin E is also a fatty-soluble vitamin that exists in 8 different isoforms with powerful antioxidant activity, the near active being α-tocopherol [98]. Numerous studies accept shown that cattle finished on pasture produce college levels of α-tocopherol in the final meat product than cattle fed high concentrate diets[23, 28, 94, 97, 99–101] (see Table 4).
Antioxidants such as vitamin E protect cells against the effects of free radicals. Free radicals are potentially damaging by-products of metabolism that may contribute to the development of chronic diseases such as cancer and cardiovascular disease.
Preliminary research shows vitamin E supplementation may help prevent or delay coronary centre disease [102–105]. Vitamin E may besides cake the formation of nitrosamines, which are carcinogens formed in the tum from nitrates consumed in the diet. It may also protect confronting the development of cancers past enhancing immune function [106]. In addition to the cancer fighting furnishings, there are some observational studies that constitute lens clarity (a diagnostic tool for cataracts) was ameliorate in patients who regularly used vitamin Eastward [107, 108]. The current recommended intake of vitamin E is 22 IU (natural source) or 33 IU (synthetic source) for men and women [93, 109], respectively, which is equivalent to fifteen milligrams by weight.
The concentration of natural α-tocopherol (vitamin E) found in grain-fed beef ranged between 0.75 to ii.92 μg/g of muscle whereas pasture-fed beefiness ranges from 2.1 to vii.73 μg/g of tissue depending on the type of forage made available to the animals (Table iv). Grass finishing increases α-tocopherol levels 3-fold over grain-fed beefiness and places grass-fed beef well within range of the musculus α-tocopherol levels needed to extend the shelf-life of retail beef (3 to 4 μg α-tocopherol/gram tissue) [110]. Vitamin E (α-tocopherol) acts post-mortem to filibuster oxidative deterioration of the meat; a process past which myoglobin is converted into chocolate-brown metmyoglobin, producing a darkened, brown appearance to the meat. In a study where grass-fed and grain-fed beef were direct compared, the brilliant red color associated with oxymyoglobin was retained longer in the retail display in the grass-fed group, even thought the grass-fed meat contains a higher concentration of more oxidizable n-3 PUFA. The authors concluded that the antioxidants in grass probably caused higher tissue levels of vitamin Due east in grazed animals with benefits of lower lipid oxidation and amend color retentiveness despite the greater potential for lipid oxidation[111].
Review of antioxidant enzyme content in grass-fed beefiness
Glutathione (GT), is a relatively new poly peptide identified in foods. It is a tripeptide composed of cysteine, glutamic acrid and glycine and functions as an antioxidant primarily as a component of the enzyme system containing GT oxidase and reductase. Inside the cell, GT has the capability of quenching free radicals (similar hydrogen peroxide), thus protecting the cell from oxidized lipids or proteins and prevent damage to DNA. GT and its associated enzymes are found in nigh all establish and brute tissue and is readily absorbed in the pocket-size intestine[112].
Although our knowledge of GT content in foods is notwithstanding somewhat limited, dairy products, eggs, apples, beans, and rice contain very piffling GT (< 3.iii mg/100 k). In contrast, fresh vegetables (eastward.one thousand., asparagus 28.three mg/100 k) and freshly cooked meats, such as ham and beef (23.three mg/100 g and 17.5 mg/100 g, respectively), are high in GT [113].
Because GT compounds are elevated in lush green forages, grass-fed beefiness is particularly high in GT equally compared to grain-fed contemporaries. Descalzo et al. (2007) reported a meaning increase in GT tooth concentrations in grass-fed beef [114]. In addition, grass-fed samples were also college in superoxide dismutase (SOD) and catalase (CAT) activity than beef from grain-fed animals[115]. Superoxide dismutase and catalase are coupled enzymes that work together as powerful antioxidants, SOD scavenges superoxide anions by forming hydrogen peroxide and CAT so decomposes the hydrogen peroxide to H2O and O2. Grass merely diets ameliorate the oxidative enzyme concentration in beef, protecting the muscle lipids against oxidation likewise equally providing the beef consumer with an additional source of antioxidant compounds.
Issues related to flavor and palatability of grass-fed beefiness
Maintaining the more favorable lipid profile in grass-fed beef requires a high percentage of lush fresh provender or grass in the ration. The higher the concentration of fresh light-green forages, the higher the αLA precursor that will exist available for CLA and northward-3 synthesis [53, 54]. Fresh pasture forages have 10 to 12 times more C18:3 than cereal grains [116]. Dried or cured forages, such every bit hay, will have a slightly lower amount of precursor for CLA and n-iii synthesis. Shifting diets to cereal grains will cause a pregnant change in the FA contour and antioxidant content within 30 days of transition [57].
Because grass-finishing alters the biochemistry of the beef, odour and season will also be affected. These attributes are directly linked to the chemical makeup of the terminal product. In a study comparing the flavor compounds betwixt cooked grass-fed and grain-fed beefiness, the grass-fed beef contained higher concentrations of diterpenoids, derivatives of chlorophyll call phyt-one-ene and phyt-2-ene, that changed both the flavour and olfactory property of the cooked product [117]. Others have identified a "green" odor from cooked grass-fed meat associated with hexanals derived from oleic and αLA FAs. In contrast to the "green" aroma, grain-fed beef was described as possessing a "soapy" aroma, presumably from the octanals formed from LA that is found in loftier concentration in grains [118]. Grass-fed beef consumers can expect a unlike season and aroma to their steaks as they cook on the grill. Likewise, because of the lower lipid content and high concentration of PUFAs, cooking time will be reduced. For an exhaustive look at the effect of meat compounds on season, see Calkins and Hodgen (2007) [119].
With respect to palatability, grass-fed beefiness has historically been less well accepted in markets where grain-fed products predominant. For example, in a study where British lambs fed grass and Spanish lambs fed milk and concentrates were assessed by British and Spanish taste panels, both found the British lamb to accept a higher scent and flavor intensity. Notwithstanding, the British panel preferred the flavor and overall eating quality of the grass-fed lamb, the Spanish panel much preferred the Castilian fed lamb [120]. Likewise, the U.Due south. is well known for producing corn-fed beef, taste panels and consumers who are more familiar with the taste of corn-fed beef seem to adopt it as well [16]. An private normally comes to prefer the foods they grew up eating, making consumer sensory panels more of an fine art than science [36]. Trained taste panels, i.eastward., persons specifically trained to evaluate sensory characteristics in beef, found grass-fed beef less palatable than grain-fed beef in flavor and tenderness [119, 121].
Conclusion
Enquiry spanning three decades supports the argument that grass-fed beefiness (on a g/m fat basis), has a more desirable SFA lipid profile (more than C18:0 cholesterol neutral SFA and less C14:0 & C16:0 cholesterol elevating SFAs) as compared to grain-fed beef. Grass-finished beef is also higher in total CLA (C18:2) isomers, TVA (C18:1 t11) and n-iii FAs on a g/yard fatty basis. This results in a meliorate n-vi:n-3 ratio that is preferred past the nutritional community. Grass-fed beefiness is besides higher in precursors for Vitamin A and E and cancer fighting antioxidants such equally GT and SOD activity equally compared to grain-fed contemporaries.
Grass-fed beef tends to be lower in overall fat content, an important consideration for those consumers interested in decreasing overall fat consumption. Because of these differences in FA content, grass-fed beefiness too possesses a distinct grass flavor and unique cooking qualities that should be considered when making the transition from grain-fed beef. To maximize the favorable lipid profile and to guarantee the elevated antioxidant content, animals should be finished on 100% grass or pasture-based diets.
Grain-fed beef consumers may achieve like intakes of both n-3 and CLA through consumption of higher fat portions with higher overall palatability scores. A number of clinical studies have shown that today'southward lean beef, regardless of feeding strategy, can be used interchangeably with fish or skinless chicken to reduce serum cholesterol levels in hypercholesterolemic patients.
Abbreviations
- c:
-
cis
- t:
-
trans
- FA:
-
fatty acid
- SFA:
-
saturated fatty acid
- PUFA:
-
polyunsaturated fatty acid
- MUFA:
-
monounsaturated fat acrid
- CLA:
-
conjugated linoleic acrid
- TVA:
-
trans-vaccenic acid
- EPA:
-
eicosapentaenoic acid
- DPA:
-
docosapentaenoic acid
- DHA:
-
docosahexaenoic acid
- GT:
-
glutathione
- SOD:
-
superoxide dismutase
- True cat:
-
catalase.
References
-
Griel AE, Kris-Etherton PM: Across saturated fat: The importance of the dietary fatty acid profile on cardiovascular disease. Nutrition Reviews. 2006, 64 (v): 257-62. 10.1111/j.1753-4887.2006.tb00208.x.
-
Kris-Etherton PM, Innis Due south: Dietary Fat Acids -- Position of the American Dietetic Clan and Dietitians of Canada. American Dietetic Association Position Report. Periodical of the American Dietetic Association. 2007, 107 (9): 1599-1611. Ref Type: Written report
-
Hu FB, Stampfer MJ, Manson JE, Rimm E, Colditz GA, Rosner BA, Hennekins CH, Willett WC: Dietary fat intake and the risk of coronary eye disease in women. New England Journal of Medicine. 1997, 337: 1491-9. 10.1056/NEJM199711203372102.
-
Posner BM, Cobb JL, Belanger AJ, Cupples LA, D'Agostino RB, Stokes J: Dietary lipid predictors of coronary heart disease in men. The Framingham Report. Archives of Internal Medicine. 1991, 151: 1181-7. 10.1001/archinte.151.6.1181.
-
Mensink RP, Katan MB: Issue of dietary fat acids on serum lipids and lipoproteins. Arteriosclerosis Thrombosis Vascular Biology. 1992, 12: 911-9.
-
Keys A: Coronary middle affliction in seven countries. Circulation. 1970, 41 (i): 211-
-
Mensink RP, Zock PL, Kester Advertising, Katan MB: Effects of dietary fatty acids and carbohydrates on the ratio of serum full HDL cholesterol and on serum lipids and apolipoproteins: A meta-analysis of 60 controlled trials. American Periodical of Clinical Nutrition. 2003, 77: 1146-55.
-
Putnam J, Allshouse J, Scott-Kantor L: U.S. per capita nutrient supply trends: More calories, refined carbohydrates, and fats. Food Review. 2002, 25 (iii): 2-15.
-
Kris-Etherton PMYS: Individual fat acid effects on plasma lipids and lipoproteins. Man studies. American Journal of Clinical Nutrition. 1997, 65 (suppl.five): 1628S-44S.
-
Higgs JD: The changing nature of red meat: 20 years improving nutritional quality. Trends in Food Science and Technology. 2000, 11: 85-95. ten.1016/S0924-2244(00)00055-viii.
-
O'Dea K, Traianedes 1000, Chisholm G, Leyden H, Sinclair AJ: Cholesterol-lowering result of a depression-fatty diet containing lean beefiness is reversed by the addition of beef fatty. American Journal of Clinical Nutrition. 1990, 52: 491-4.
-
Beauchesne-Rondeau E, Gascon A, Bergeron J, Jacques H: Plasma lipids and lipoproteins in hypercholesterolemic men fed a lipid-lowering diet containing lean beef, lean fish, or poultry. American Journal of Clinical Nutrition. 2003, 77 (3): 587-93.
-
Melanson K, Gootman J, Myrdal A, Kline K, Rippe JM: Weight loss and total lipid profile changes in overweight women consuming beef or craven equally the primary protein source. Diet. 2003, 19: 409-14. 10.1016/S0899-9007(02)01080-8.
-
Denke MA: Role of beef and beefiness tallow, an enriched source of stearic acid, in a cholesterol-lowering diet. American Journal of Clinical Nutrition. 1994, threescore: 1044S-9S.
-
Smith DR, Wood R, Tseng Due south, Smith SB: Increased beef consumption increases lipoprotein A-I merely not serum cholesterol of mildly hypercholesterolemic men with different levels of habitual beefiness intake. Experimental Biological Medicine. 2002, 227 (four): 266-75.
-
Wood JD, Richardson RI, Nute GR, Fisher AV, Campo MM, Kasapidou E, Sheard PR, Enser M: Furnishings of fat acids on meat quality: review. Meat Scientific discipline. 2003, 66: 21-32. 10.1016/S0309-1740(03)00022-half-dozen.
-
Williamson CS, Foster RK, Stanner SA, Buttriss JL: Crimson meat in the diet. British Diet Foundation. Nutrition Bulletin. 2005, 30: 323-335. ten.1111/j.1467-3010.2005.00525.x. Ref Blazon: Written report
-
Biesalski HK: Meat as a component of a good for you diet - are there any risks or benefits if meat is avoided?. Meat Science. 2005, 70 (3): 509-24. 10.1016/j.meatsci.2004.07.017.
-
Yu Southward, Derr J, Etherton TD, Kris-Etherton PM: Plasma cholesterol-predictive equations demonstrate that stearic acid is neutral and monosaturated fatty acids are hypocholesterolemic. American Journal of Clinical Diet. 1995, 61: 1129-39.
-
Whetsell MS, Rayburn EB, Lozier JD: Human being Health Effects of Fatty Acids in Beef. 2003, Fact Sail: West Virgina University & U.Southward.D.A. Agriculture Enquiry Service. Extension Service Westward Virginia Academy, Ref Type: Electronic Citation
-
Kris-Etherton PM: Monounsaturated fatty acids and run a risk of cardiovascular disease. Circulation. 1999, 100: 1253-
-
DeSmet Due south, Raes K, Demeyer D: Meat fatty acid composition as affected by fatness and genetic factors: a review. Animal Enquiry. 2004, 53: 81-98. ten.1051/animres:2004003.
-
De la Fuente J, Diaz MT, Alvarez I, Oliver MA, Font i Furnols M, Sanudo C, Campo MM, Montossi F, Nute GR, Caneque 5: Fatty acid and vitamin Due east composition of intramuscular fat in cattle reared in different production systems. Meat Scientific discipline. 2009, 82: 331-7. 10.1016/j.meatsci.2009.02.002.
-
Garcia PT, Pensel NA, Sancho AM, Latimori NJ, Kloster AM, Amigone MA, Casal JJ: Beef lipids in relation to animal breed and nutrition in Argentine republic. Meat Science. 2008, 79: 500-8. 10.1016/j.meatsci.2007.10.019.
-
Alfaia CPM, Alves SP, Martins SIV, Costa ASH, Fontes CMGA, Lemos JPC, Bessa RJB, Prates JAM: Event of feeding system on intramuscular fat acids and conjugated linoleic acid isomers of beefiness cattle, with accent on their nutritional value and discriminatory power. Food Chemical science. 2009, 114: 939-46. 10.1016/j.foodchem.2008.10.041.
-
Leheska JM, Thompson LD, Howe JC, Hentges East, Boyce J, Brooks JC, Shriver B, Hoover L, Miller MF: Effects of conventional and grass-feeding systems on the nutrient limerick of beefiness. Journal Animal Science. 2008, 86: 3575-85. 10.2527/jas.2007-0565.
-
Nuernberg K, Dannenberger D, Nuernberg G, Ender K, Voigt J, Scollan ND, Wood JD, Nute GR, Richardson RI: Consequence of a grass-based and a concentrate feeding system on meat quality characteristics and fatty acid limerick of longissimus musculus in different cattle breeds. Livestock Production Science. 2005, 94: 137-47. x.1016/j.livprodsci.2004.11.036.
-
Realini CE, Duckett SK, Brito GW, Rizza MD, De Mattos D: Result of pasture vs. concentrate feeding with or without antioxidants on carcass characteristics, fatty acid composition, and quality of Uruguayan beef. Meat Scientific discipline. 2004, 66: 567-77. 10.1016/S0309-1740(03)00160-viii.
-
Warren HE, Enser Grand, Richardson I, Woods JD, Scollan ND: Result of breed and diet on total lipid and selected shelf-life parameters in beef musculus. Proceedings of British Club of animal science. 2003, 23-
-
Ponnampalam EN, Mann NJ, Sinclair AJ: Effect of feeding systems on omega-3 fatty acids, conjugated linoleic acid and trans fat acids in Australian beef cuts, potential touch on human health. Asia Pacific Journal of Clinical Diet. 2006, 15 (i): 21-nine.
-
Descalzo A, Insani EM, Biolatto A, Sancho AM, Garcia PT, Pensel NA: Influence of pasture or grain-based diets supplemented with vitamin E on antioxidant/oxidative balance of Argentine beefiness. Meat Science. 2005, 70: 35-44. 10.1016/j.meatsci.2004.11.018.
-
Wheeler TL, Davis GW, Stoecker BJ, Harmon CJ: Cholesterol concentrations of longissimus musculus, subcutaneous fat and serum of two beef cattle breed types. Journal of Animal Science. 1987, 65: 1531-vii.
-
Smith DR, Wood R, Tseng S, Smith SB: Increased beef consumption increases apolipoprotein A-ane merely non serum cholesterol of mildly hypercholesterolemic men with different levels of habitual beef intake. Experimental Biological Medicine. 2002, 227 (iv): 266-75.
-
Dominion DC, Broughton KS, Shellito SM, Maiorano G: Comparison of muscle fatty acid profiles and cholesterol concentrations of bison, cattle, elk and craven. Journal Brute Scientific discipline. 2002, 80: 1202-11.
-
Alfaia CPM, Castro MLF, Martins SIV, Portugal APV, Alves SPA, Fontes CMGA: Influence of slaughter flavor and muscle type on faty acrid composition, conjugated linoleic acid isomeric distribution and nutritional quality of intramuscular fat in Arouquesa-PDO veal. Meat Science. 2007, 76: 787-95. 10.1016/j.meatsci.2007.02.023.
-
Sitz BM, Calkins CR, Feuz DM, Umberger WJ, Eskridge KM: Consumer sensory acceptance and value of domestic, Canadian, and Australian grass-fed beef steaks. Journal of Animal Scientific discipline. 2005, 83: 2863-8.
-
Bauman DE, Lock AL: Conjugated linoleic acid: biosynthesis and nutritional significance. Fox and McSweeney. Advanced Dairy Chemistry. 2006, Springer, New York, 93-136. Ref Type: Serial (Volume, Monograph), three, two
-
Enser M, Hallett KG, Hewett B, Fursey GAJ, Wood JD, Harrington G: Fatty acid content and limerick of UK beefiness and lamb muscle in relation to production system and implications for human nutrition. Meat Science. 1998, 49 (3): 329-41. 10.1016/S0309-1740(97)00144-7.
-
Ruxton CHS, Reed SC, Simpson JA, Millington KJ: The wellness benefits of omega-iii polyunsaturated fat acids: a review of the evidence. The Periodical of Human being Nutrition and Dietetics. 2004, 17: 449-59. 10.1111/j.1365-277X.2004.00552.x.
-
Simopoulos A: Omega-iii fatty acids in health and affliction and in growth and evolution. American Periodical of Clinical Diet. 1991, 54: 438-63.
-
Thomas BJ: Efficiency of conversion of alpha-linolenic acrid to long concatenation n-three fatty acids in human. Current Stance in Clincal Nutrition and Metabolic Intendance. 2002, 5 (ii): 127-32. 10.1097/00075197-200203000-00002.
-
Connor WE: Importance of n-3 fat acids in health and disease. American Journal of Clinical Diet. 2000, 71: 171S-5S.
-
Kremer JM, Lawrence DA, Jubiz W, Galli C, Simopoulos AP: Unlike doses of fish -oil fatty acid ingestion in active rheumatoid arthritis: a prospective study of clinical and immunological parameters. Dietary Omega-3 and Omega-half dozen fatty acids: biological effects and nutritional essentiality. 1989, New York: Plenum Printing
-
DiGiacomo RA, Kremer JM, Shah DM: Fish-oil dietary supplementation in patients with Raynaud's Phenomenon: A double-blind, controlled, prospective study. The American Journal of Medicine. 1989, 86: 158-64. 10.1016/0002-9343(89)90261-one.
-
Kalmijn South: Dietary fat intake and the risk of incident dementia in the Rotterdam Report. Register of Neurology. 1997, 42 (5): 776-82. ten.1002/ana.410420514.
-
Yehuda S, Rabinovtz Southward, Carasso RL, Mostofsky DI: Essential fatty acids preparation (SR-3) improves Alzheimer's patient's quality of life. International Journal of Neuroscience. 1996, 87 (3-4): 141-9. 10.3109/00207459609070833.
-
Hibbeln JR: Fish oil consumption and major depression. The Lancet. 1998, 351: 1213-10.1016/S0140-6736(05)79168-half dozen. (April 18 1998)
-
Hibbeln JR, Salem N: Dietary polyunsaturated fatty acids and low: when cholesterol does not satisfy. American Journal of Clinical Nutrition. 1995, 62: 1-nine.
-
Stoll AL, et al: Omega 3 fat acids in bipolar disorder. Archives of General Psychiatry. 1999, 56: 407-12-415-sixteen
-
Calabrese JR, Rapport DJ, Shleton Medico: Fish oils and bipolar disorder. Archives of Full general Psychiatry. 1999, 56: 413-4. 10.1001/archpsyc.56.five.413.
-
Laugharne JDE: Fatty acids and schizophrenia. Lipids. 1996, 31: S163-S165. 10.1007/BF02637070.
-
Sinclair AJ, Johnson L, O'Dea K, Holman RT: Diets rich in lean beef increase arachidonic acrid and long-chain omega 3 polyunsaturated fatty acid levels in plasma phospholipids. Lipids. 1994, 29 (5): 337-43. 10.1007/BF02537187.
-
Raes K, DeSmet Southward, Demeyer D: Event of dietary fat acids on incorporation of long chain polyunsaturated fat acids and conjugated linoleic acid in lamb, beefiness and pork meat: a review. Animal Feed Science and Technology. 2004, 113: 199-221. 10.1016/j.anifeedsci.2003.09.001.
-
Marmer WN, Maxwell RJ, Williams JE: Effects of dietary regimen and tissue site on bovine fat acid profiles. Periodical Animal Science. 1984, 59: 109-21.
-
Wood JD, Enser Grand: Factors influencing fatty acids in meat and the role of antioxidants in improving meat quality. British Journal of Nutrition. 1997, 78: S49-S60. x.1079/BJN19970134.
-
French P, Stanton C, Lawless F, O'Riordan EG, Monahan FJ, Caffery PJ, Moloney AP: Fatty acrid composition, including conjugated linoleic acid of intramuscular fat from steers offered grazed grass, grass silage or concentrate-based diets. Journal Animate being Science. 2000, 78: 2849-55.
-
Duckett SK, Wagner DG, Yates LD, Dolezal HG, May SG: Effects of fourth dimension on feed on beefiness food composition. Journal Animal Science. 1993, 71: 2079-88.
-
Nuernberg K, Nuernberg Thousand, Ender K, Lorenz S, Winkler K, Rickert R, Steinhart H: Omega-iii fat acids and conjugated linoleic acids of longissimus muscle in beef cattle. European Periodical of Lipid Scientific discipline Technology. 2002, 104: 463-71. 10.1002/1438-9312(200208)104:8<463::Assist-EJLT463>3.0.CO;2-U.
-
Griinari JM, Corl BA, Lacy SH, Chouinard PY, Nurmela KV, Bauman DE: Conjugated linoleic acid is synthesized endogenoulsy in lactating dairy cows by delta-ix desaturase. Journal of Nutrition. 2000, 130: 2285-91.
-
Sehat Northward, Rickert RR, Mossoba MM, Dramer JKG, Yurawecz MP, Roach JAG, Adlof RO, Morehouse KM, Fritsche J, Eulitz KD, Steinhart H, Ku K: Improved separation of conjugated fat acrid methyl esters past argent ion-high-performance liquid chromatography. Lipids. 1999, 34: 407-xiii. x.1007/s11745-999-0379-3.
-
Pariza MW, Park Y, Cook ME: Mechanisms of action of conjugated linoleic acrid: evidence and speculation. Proceedings for the Society of Experimental Biological science and Medicine. 2000, 32: 853-8.
-
Bessa RJB, Santos-Silva J, Ribeiro JMR, Portugal AV: Reticulo-rumen biohydrogenation and the enrichment of ruminant edible products with linoleic acrid conjugated isomers. Livestock Production Science. 2000, 63: 201-11. 10.1016/S0301-6226(99)00117-7.
-
Turpeinen AM, Mutanen M, Aro ASI, Basu SPD, Griinar JM: Bioconversion of vaccenic acrid to conjugated linoleic acid in humans. American Journal of Clinical Diet. 2002, 76: 504-10.
-
Turpeinen AM, Mautanen G, Aro A, Salminen I, Basu South, Palmquist DL: Bioconversion of vaccenic acid to conjugated linoleic acid in humans. American Journal of Clinical Nutrition. 2002, 76: 504-10.
-
Turpeinen AM, Mautanen M, Aro A, Salminen I, Basu S, Palmquist DL: Bioconversion of vaccenic acid to conjugated linoleic acid in humans. American Journal of Clinical Nutrition. 2002, 76: 504-10.
-
Adlof RO, Duval S, Emken EA: Biosynthesis of conjugated linoleic acid in humans. Lipids. 2000, 35: 131-5. 10.1007/BF02664761.
-
Mandell IB, Gullett JG, Buchanan-Smith JG, Campbell CP: Effects of diet and slaughter endpoint on carcass limerick and beef quality in Charolais cantankerous steers fed alfalfa silage and (or) loftier concentrate diets. Canadian Journal of Animal Science. 1997, 77: 403-14.
-
Dugan MER, Rollan DC, Aalhus JL, Aldai N, Kramer JKG: Subcutaneous fat limerick of youthful and mature Canadian beefiness: emphasis on private conjugated linoleic acrid and trans-18:1 isomers. Canadian Journal of Brute Science. 2008, 88: 591-ix.
-
Hodgson JM, Wahlqvist ML, Boxall JA, Balazs ND: Platelet trans fatty acids in relation to angiographically assessed coronary avenue disease. Atherosclerosis. 1996, 120: 147-54. 10.1016/0021-9150(95)05696-3.
-
IP C, Scimeca JA, Thompson HJ: Conjugated linoleic acrid. Cancer Supplement. 1994, 74 (3): 1050-4.
-
Kritchevsky D, Tepper SA, Wright S, Tso P, Czarnecki SK: Influence of conjugated linoleic acid (CLA) on institution and progression of atherosclerosis in rabbits. Journal American Collection of Diet. 2000, 19 (4): 472S-7S.
-
Steinhart H, Rickert R, Winkler K: Identification and assay of conjugated linoleic acid isomers (CLA). European Journal of Medicine. 1996, 20 (8): 370-2.
-
Dugan MER, Aalhus JL, Jeremiah LE, Kramer JKG, Schaefer AL: The effects of feeding conjugated linoleic acid on subsequent port quality. Canadian Periodical of Creature Science. 1999, 79: 45-51.
-
Park Y, Albright KJ, Liu Westward, Storkson JM, Melt ME, Pariza MW: Effect of conjugated linoleic acid on body composition in mice. Lipids. 1997, 32: 853-eight. x.1007/s11745-997-0109-x.
-
Sisk M, Hausman D, Martin R, Azain M: Dietary conjugated linoleic acid reduces adiposity in lean simply not obese Zucker rats. Journal of Nutrition. 2001, 131: 1668-74.
-
Smedman A, Vessby B: Conjugated linoleic acid supplementation in humans - Metabolic effects. Periodical of Diet. 2001, 36: 773-81.
-
Tsuboyama-Kasaoka N, Takahashi G, Tanemura K, Kim HJ, Tange T, Okuyama H, Kasai One thousand, Ikemoto SS, Ezaki O: Conjugated linoleic acid supplementation reduces adipose tissue by apoptosis and develops lipodystrophy in mice. Diabetes. 2000, 49: 1534-42. 10.2337/diabetes.49.9.1534.
-
Clement L, Poirier H, Niot I, Bocher V, Guerre-Millo Chiliad, Krief B, Staels B, Besnard P: Dietary trans-10, cis-12 conjugated linoleic acid induces hyperinsulemia and fatty liver in the mouse. Journal of Lipid Research. 2002, 43: 1400-9. 10.1194/jlr.M20008-JLR200.
-
Roche HM, Noone E, Sewter C, McBennett Southward, Savage D, Gibney MJ, O'Rahilly S, Vidal-Plug AJ: Isomer-dependent metabolic furnishings of conjugated linoleic acid: insights from molecular markers sterol regulatory chemical element-bounden protein 1c and LXR blastoff. Diabetes. 2002, 51: 2037-44. x.2337/diabetes.51.vii.2037.
-
Riserus U, Arner P, Brismar K, Vessby B: Handling with dietary trans x cis 12 conjugated linoleic acrid causes isomer specific insulin resistance in obese men with the metabolic syndrome. Diabetes Care. 2002, 25: 1516-21. ten.2337/diacare.25.9.1516.
-
Delany JP, Blohm F, Truett AA, Scimeca JA, West DB: Conjugated linoleic acid rapidly reduces trunk fatty content in mice without affecting energy intake. American Journal of Physiology. 1999, 276 (4 pt 2): R1172-R1179.
-
Kelley DS, Simon VA, Taylor PC, Rudolph IL, Benito P: Dietary supplementation with conjugated linoleic acid increased its concentration in human peripheral blood mononuclear cells, only did not alter their role. Lipids. 2001, 36: 669-74. x.1007/s11745-001-0771-z.
-
Whigham LD, Cook ME, Atkinson RL: Conjugated linoleic acid: Implications for human health. Pharmacological Inquiry. 2000, 42 (6): 503-10. 10.1006/phrs.2000.0735.
-
Schmid A, Collomb Thou, Sieber R, Bee G: Conjugated linoleic acid in meat and meat products. A review Meat Science. 2006, 73: 29-41. 10.1016/j.meatsci.2005.x.010.
-
Knekt P, Jarvinen R, Seppanen R, Pukkala E, Aromaa A: Intake of dairy products and the risk of breast cancer. British Periodical of Cancer. 1996, 73: 687-91.
-
Ha YL, Grimm NK, Pariza MW: Newly recognized anticarcinogenic fatty acids: identification and quantification in natural and candy cheese. Journal of Agronomical and Food Chemical science. 1989, 37: 75-81. 10.1021/jf00085a018.
-
Ritzenthaler KL, McGuire MK, Falen R, Shultz TD, Dasgupta North, McGuire MA: Interpretation of conjugated linoleic acid intake by written dietary assessment methodologies underestimates bodily intake evaluated by food duplicate methodology. Journal of Nutrition. 2001, 131: 1548-54.
-
Parodi PW: Conjugated linoleic acid: an anticarcinogenic fatty acid present in milk fatty (review). Australian Journal of Dairy Engineering. 1994, 49 (2): 93-7.
-
Dunne PG, Monahan FJ, O'Mara FP, Moloney AP: Colour of bovine subcutaneous adipose tissue: A review of contributory factors, associations with carcass and meat quality and its potential utility in authentication of dietary history. Meat Science. 2009, 81 (1): 28-45. 10.1016/j.meatsci.2008.06.013.
-
Chauveau-Duriot B, Thomas D, Portelli J, Doreau M: Carotenoids content in forages: variation during conservation. Renc Rech Ruminants. 2005, 12: 117-
-
Scott LW, Dunn JK, Pownell HJ, Brauchi DJ, McMann MC, Herd JA, Harris KB, Savell JW, Cross Hour, Gotto AM: Furnishings of beef and craven consumption on plasma lipid levels in hypercholesterolemic men. Archives of Internal Medicine. 1994, 154 (11): 1261-7. 10.1001/archinte.154.xi.1261.
-
Hunninghake DB, Maki KC, Kwiterovick PO, Davidson MH, Dicklin MR, Kafonek SD: Incorporation of lean carmine meat National Cholesterol Educational activity Program Step I diet: a long-term, randomized clinical trial in free-living persons with hypercholesterolemic. Journal of American Colleges of Diet. 2000, xix (iii): 351-60.
-
National Institute of Health Clinical Diet Center: Facts nigh dietary supplements: Vitamin A and Carotenoids. 2002, Ref Blazon: Pamphlet
-
Descalzo AM, Insani EM, Biolatto A, Sancho AM, Garcia PT, Pensel NA, Josifovich JA: Influence of pasture or grain-based diets supplemented with vitamin E on antioxidant/oxidative rest of Argentine beef. Journal of Meat Scientific discipline. 2005, 70: 35-44. 10.1016/j.meatsci.2004.xi.018.
-
Simonne AH, Green NR, Bransby DI: Consumer acceptability and beta-carotene content of beef as related to cattle finishing diets. Periodical of Food Scientific discipline. 1996, 61: 1254-6. 10.1111/j.1365-2621.1996.tb10973.x.
-
Duckett SK, Pratt SL, Pavan Due east: Corn oil or corn grain supplementation to stters grazing endophyte-free alpine fescue. II. Effects on subcutaneous fatty acid content and lipogenic gene expression. Journal of Animal Science. 2009, 87: 1120-8. 10.2527/jas.2008-1420.
-
Yang A, Brewster MJ, Lanari MC, Tume RK: Effect of vitamin E supplementation on alpha-tocopherol and beta-carotene concentrations in tissues from pasture and grain-fed cattle. Meat Scientific discipline. 2002, 60 (ane): 35-twoscore. 10.1016/S0309-1740(01)00102-four.
-
Pryor WA: Vitamin Due east and Carotenoid Abstracts- 1994 Studies of Lipid-Soluble Antioxidants. Vitamin Eastward Enquiry and Information Services. 1996
-
Arnold RN, Scheller N, Arp KK, Williams SC, Beuge DR, Schaefer DM: Effect of long or short-term feeding of alfa-tocopherol acetate to Holstein and crossbred beef steers on performance, carcass characteristics, and beef color stability. Periodical Creature Science. 1992, lxx: 3055-65.
-
Descalzo AM, Sancho AM: A review of natural antioxidants and their effects on oxidative status, odor and quality of fresh beefiness in Argentina. Meat Science. 2008, 79: 423-36. 10.1016/j.meatsci.2007.12.006.
-
Insani EM, Eyherabide A, Grigioni G, Sancho AM, Pensel NA, Descalzo AM: Oxidative stability and its relationship with natural antioxidants during refrigerated retail display of beef produced in Argentine republic. Meat Science. 2008, 79: 444-52. 10.1016/j.meatsci.2007.x.017.
-
Lonn EM, Yusuf S: Is there a role for antioxidant vitamins in the prevention of cardiovascular diseases? An update on epidemiological and clinical trials data. Cancer Journal of Cardiology. 1997, xiii: 957-65.
-
Jialal I, Fuller CJ: Result of vitamin Due east, vitamin C and beta-carotene on LDL oxidation and atherosclerosis. Canadian Periodical of Cardiology. 1995, eleven (supplemental M): 97G-103G.
-
Stampfer MJ, Hennekens CH, Manson JE, Colditz GA, Rosner B, Willett WC: Vitamin E consumption and the take chances of coronary disease in women. New England Journal of Medicine. 1993, 328 (1444): 1449-
-
Knekt P, Reunanen A, Jarvinen R, Seppanen R, Heliovaara Chiliad, Aromaa A: Antioxidant vitamin intake and coronary bloodshed in a longitudinal population study. American Journal of Epidemiology. 1994, 139: 1180-9.
-
Weitberg AB, Corvese D: Effects of vitamin E and beta-carotene on DNA strand breakage induced by tobacco-specific nitrosamines and stimulated homo phagocytes. Journal of Experimental Cancer Inquiry. 1997, 16: eleven-4.
-
Leske MC, Chylack LT, He Q, Wu SY, Schoenfeld E, Friend J, Wolfe J: Antioxidant vitamins and nuclear opacities: The longitudinal study of cataract. Ophthalmology. 1998, 105: 831-vi. 10.1016/S0161-6420(98)95021-7.
-
Teikari JM, Virtamo J, Rautalahi Thousand, Palmgren J, Liestro M, Heinonen OP: Long-term supplementation with alpha-tocopherol and beta-carotene and historic period-related cataract. Acta Ophthalmologica Scandinavica. 1997, 75: 634-40. 10.1111/j.1600-0420.1997.tb00620.x.
-
Dietary guidelines Advisory Committee, Agricultural Inquiry Service United States Department of Agriculture USDA: Report of the dietary guidelines advisory committee on the dietary guidelines for Americans. Dietary guidelines Informational Committee. 2000, Ref Type: Hearing
-
McClure EK, Belk KE, Scanga JA, Smith GC: Decision of appropriate Vitamin E supplementation levels and administration times to ensure adequate muscle tissue alpha-tocopherol concentration in cattle destined for the Nolan Ryan tender-aged beef program. Fauna Sciences Inquiry Report. 2002, The Section of Animal Sciences, Colorado State University, Ref Type: Study
-
Yang A, Lanari MC, Brewster MJ, Tume RK: Lipid stability and meat colour of beef from pasture and grain-fed cattle with or without vitamin E supplement. Meat Scientific discipline. 2002, 60: 41-50. x.1016/S0309-1740(01)00103-half-dozen.
-
Valencia E, Marin A, Hardy 1000: Glutathione - Nutritional and Pharmacological Viewpoints: Part Two. Nutraceuticals. 2001, 17: 485-half dozen.
-
Valencia E, Marin A, Hardy G: Glutathione - Nutritional and Pharmacologic Viewpoints: Part IV. Nutraceuticals. 2001, 17: 783-4.
-
Descalzo AM, Rossetti L, Grigioni G, Irurueta M, Sancho AM, Carrete J, Pensel NA: Antioxidant status and odor contour in fresh beef from pasture or grain-fed cattle. Meat Science. 2007, 75: 299-307. 10.1016/j.meatsci.2006.07.015.
-
Gatellier P, Mercier Y, Renerre M: Effect of diet finishing fashion (pasture or mixed nutrition) on antioxidant status of Charolais bovine meat. Meat Science. 2004, 67: 385-94. 10.1016/j.meatsci.2003.11.009.
-
French P, O'Riordan EG, Monahan FJ, Caffery PJ, Moloney AP: Fatty acrid composition of intra-muscular tricylglycerols of steers fed autumn grass and concentrates. Livestock Production Science. 2003, 81: 307-17. 10.1016/S0301-6226(02)00253-1.
-
Elmore JS, Warren HE, Mottram DS, Scollan ND, Enser Thou, Richardson RI: A comparing of the aroma volatiles and fat acid compositions of grilled beefiness musculus from Aberdeen Angus and Holstein-Friesian steers fed deits based on silage or concentrates. Meat Scientific discipline. 2006, 68: 27-33. 10.1016/j.meatsci.2004.01.010.
-
Lorenz S, Buettner A, Ender K, Nuernberg M, Papstein HJ, Schieberle P: Influence of keeping system on the fatty acid composition in the longissimus muscle of bulls and odorants formed after pressure-cooking. European Food Research and Applied science. 2002, 214: 112-8. ten.1007/s00217-001-0427-4.
-
Calkins CR, Hodgen JM: A fresh look at meat flavor. Meat Science. 2007, 77: 63-80. 10.1016/j.meatsci.2007.04.016.
-
Sanudo C, Enser ME, Campo MM, Nute GR, Maria 1000, Sierra I, Wood JD: Fatty acid composition and sensory characteristics of lamb carcasses from Britain and Spain. Meat Science. 2000, 54: 339-46. 10.1016/S0309-1740(99)00108-4.
-
Killinger KM, Calkins CR, Umberger WJ, Feuz DM, Eskridge KM: A comparison of consumer sensory credence and value of domestic beefiness steaks and steaks form a branded, Argentine beef plan. Journal Fauna Science. 2004, 82: 3302-7.
Acknowledgements
The authors would similar to acknowledge Grace Berryhill for her assistance with the figures, tables and editorial contributions to this manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional data
Competing interests
The authors declare that they have no competing interests.
Authors' contributions
CAD was responsible for the literature review, completed most of the primary writing, created the manuscript and worked through the submission procedure; AA conducted the literature search, organized the manufactures according to category, completed some of the master writing and served as editor; SPD conducted a portion of the literature review and served as editor for the manuscript; GAN conducted a portion of the literature review and served every bit editor for the manuscript; SL conducted a portion o the literature review and served as editor for the manuscript. All authors read and approved the final manuscript.
Authors' original submitted files for images
Rights and permissions
This article is published under license to BioMed Central Ltd. This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in whatsoever medium, provided the original work is properly cited.
Reprints and Permissions
Most this commodity
Cite this article
Daley, C.A., Abbott, A., Doyle, P.S. et al. A review of fatty acrid profiles and antioxidant content in grass-fed and grain-fed beef. Nutr J 9, ten (2010). https://doi.org/10.1186/1475-2891-nine-10
-
Received:
-
Accepted:
-
Published:
-
DOI : https://doi.org/10.1186/1475-2891-9-10
Keywords
- Conjugated Linoleic Acid
- Conjugated Linoleic Acid Isomer
- Antioxidant Content
- Total Conjugated Linoleic Acid
- Conjugated Linoleic Acid C9t11
Source: https://nutritionj.biomedcentral.com/articles/10.1186/1475-2891-9-10
0 Response to "What Is Healthier Grass Feed Ir Organic Beef"
Postar um comentário